What Is an Implantable Venous Access Port (PORT)?
An implantable venous access port (PORT), also known as an implantable venous catheter system, is a fully implantable, closed intravenous infusion system. Approximately 80% of PORTs are used in oncology care, primarily for the administration of chemotherapy drugs. They are also widely used for intravenous infusion, parenteral nutrition, blood product transfusion, and blood sampling.

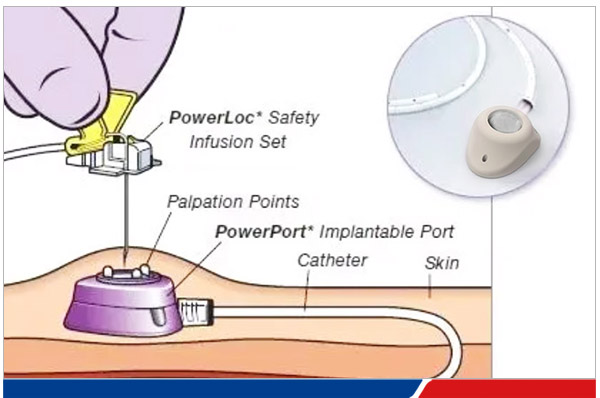
PORT systems are compatible with the infusion of medications with varying properties and are designed to provide safe, long-term venous access.
A totally implantable venous access port is completely embedded beneath the skin and intended for long-term use. The system consists of:
- A catheter positioned in the superior vena cava
- A subcutaneously implanted injection port (reservoir)
The catheter is inserted via the internal jugular vein or subclavian vein, with the port placed in the subclavian region and connected to the catheter, forming a reliable long-term venous access route.
Depending on the implantation site, PORTs can be classified as chest ports or arm ports. They are suitable for drug delivery, fluid infusion, nutritional support, blood transfusion, and blood sampling.
Implant-grade PEEK materials offer excellent physicochemical properties, radiolucency, and biocompatibility. They have been successfully used in orthopedic oncology, plastic and reconstructive surgery, spine applications, knee joints, and craniofacial reconstruction, partially replacing metal and other high-performance polymer materials.
Advantages of AKSOPEEK®
Why Choose AKSOPEEK® PEEK PORTs Over Conventional PORTs?
- Excellent biocompatibility, suitable for long-term implantation
- Low density, making the port lighter than metal-based alternatives and improving patient comfort
- Outstanding thermal insulation, reducing heat buildup during bathing or sun exposure
- Inherent antibacterial properties, helping lower infection risk and reduce complications during long-term implantation
- Superior corrosion resistance, minimizing reactions with infused medications and fluids
- High radiation resistance, ensuring performance stability during radiotherapy and chemotherapy
Implant-Grade PEEK
Implant-grade PEEK refers to PEEK materials specifically developed for use in implantable medical devices. These materials are widely applied in:
- Orthopedic implants (e.g., interbody fusion cages, ligament repair anchors, joint interface screws)
- Neurosurgical repair components (e.g., cranial and maxillofacial implants)
- Cardiovascular devices (e.g., heart valves, pacemaker housings)
In recent years, modified PEEK materials have also been increasingly used in dental implants and trauma fixation plates, where high mechanical performance is required.
Beyond standard medical-grade biocompatibility, implant-grade PEEK must meet more stringent biosafety requirements, including systemic toxicity, genotoxicity, carcinogenicity, blood compatibility, and implantation response testing. It must also comply with YY/T 0660-2008 — Polyetheretherketone (PEEK) Polymers for Surgical Implants.
AKSOPEEK® Implant-Grade PEEK Materials
AKSOPEEK® is a dedicated series of implant-grade PEEK materials developed to meet the growing demands of advanced medical implant applications. Since 2018, Junhua Medical has systematically advanced its implant-grade PEEK program, completing comprehensive third-party evaluations covering biological performance, physicochemical properties, and aging behavior.
To further standardize and expand implant-grade PEEK applications, multiple AKSOPEEK® grades have been introduced.
AKSOPEEK® Product Series
- AKSOPEEK® – Implant-grade pure PEEK
- AKSOPEEK® HA – Implant-grade PEEK enhanced with hydroxyapatite for improved biocompatibility
- AKSOPEEK® CF – Implant-grade PEEK reinforced with short carbon fibers
- AKSOPEEK® LCF – Implant-grade PEEK reinforced with continuous carbon fibers
Application Scenarios
AKSOPEEK® implant-grade PEEK materials demonstrate excellent physicochemical performance, radiolucency, biocompatibility, and mechanical compatibility with human bone. Their performance is comparable to leading international alternatives and they are widely applied in:
- Orthopedic oncology
- Plastic and reconstructive surgery
- Spine surgery
- Knee joint applications
- Craniofacial reconstruction
AKSOPEEK® materials provide a reliable solution for advanced implantable medical devices, supporting long-term safety, comfort, and clinical performance.


